In these unprecedented times, we know that controlling the spread of novel coronavirus requires social distancing and testing. While all of us are doing our parts to stay at home, there’s a lot of confusion, chaos and frustration surrounding coronavirus testing in the US.
Before going further, I want to be clear that I am not a medical doctor or researcher. I’ve been working closely with our portfolio company Scanwell Health who made a very important announcement last Friday about their work on at-home coronavirus testing. As such, I have received countless questions on COVID-19 diagnostics and thought it would be helpful to provide a high-level overview on the latest testing technology.
What type of tests are there?
There are two types of tests.
- RT-PCR-based method
This approach looks for the RNA of the virus using a technique called RT-PCR (reverse transcription polymerase chain reaction). It’s a very sensitive test and is the standard method used by healthcare professionals and hospitals in the US today.
Typically, a healthcare professional uses a nasopharyngeal swab to collect a mucus sample from a patient. Other specimens via bronchoalveolar lavage and oropharyngeal swabs can also be used. That sample is then sent to a CLIA-certified lab for molecular analysis. The result tells us whether the virus is present genetically in the patient, i.e. whether you currently have COVID-19.
When we hear about test shortages and delays, this is attributed to:
- Shortages of swabs and specific RNA extraction reagents
- Time to analyze samples. The exact time depends on the equipment used in a particular lab. For example, Cepheid can run specimens in 45 mins, but it typically takes upwards of 24 hours. Note that analysis must be done in a CLIA-certified lab
- Cost: this is an expensive test (>$100) due to the two factors above
- Serological-based method
This is a blood-based test that can be used to identify whether someone has been exposed to a particular pathogen – in this case, the SARS-CoV-2 virus.
A healthcare professional draws blood by inserting a needle into a patient’s vein. This venous blood sample is then analyzed in a test-cassette where it is combined with a diluent. After 15 minutes, colored lines will appear to indicate the presence of specific antibodies.
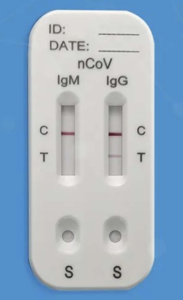
Example of a rapid serological test developed by Innovita
As of right now, researchers have not found any evidence that the natural antibodies to the SARS-CoV-2 virus exist without exposure. Therefore, if this rapid test detects the IgM and IgG antibodies in the blood, we can conclude that the person has been exposed and developed antibodies against the virus. IgM antibodies appear first (typically after 1 week exposure) and IgG appears in the serum about 2 weeks after exposure. I’ll comment more on immune response shortly.
Serological test kits can be produced at scale and at a reasonable cost (~$10).
Currently, these rapid tests are limited to professional use (i.e. performed in a lab by a technician) and have been used widely in Asia. But given the new FDA guidance, these tests can now be performed professionally in the US too without needing an Emergency Use Authorization (EUA).
Which test is better or more effective?
RT-PCR is more technically accurate when positive for an active infection, but the serological approach is much easier to administer and provides faster results. Serology tests also provide additional information about immunity (past exposure) that is not available from a RT-PCR test.
Some early studies taking the serological approach show that antibodies are present as early as three to five days for some patients. The median time for seroconversion of IgM was 12 days, and rapidly increased to 94.3% at Day 15.
“The seroconversion sequentially appeared for Ab, IgM and then IgG, with a median time of 11, 12 and 14 days, respectively. The presence of antibodies was < 40% among patients in the first 7 days of illness, and then rapidly increased to 100.0%, 94.3% and 79.8% for Ab, IgM and IgG respectively since day 15 after onset.”
In addition, based on a clinical study performed by a Chinese biotech company, Innovita, rapid serological test sensitivity was 87.3% and specificity was 100%. And while PCR tests can have higher sensitivity if performed correctly, it is highly dependent on collection method. For instance, the RT-PCR sensitivity compares as follows:
- Bronchoalveolar lavage fluid: 93%,
- Sputum: 72%,
- Nasal swabs: 63% (most common sample collection)
- Fibrobronchoscope brush biopsy: 46%
How do we act on the results of each test?
For RT-PCR, the interpretation is simple. If positive, then you are actively fighting the infection (whether you are symptomatic or asymptomatic) and will be treated accordingly.
For serological tests, it is still early to call this a true diagnostic. We need to administer more tests and collect more data to be definitive on what action to take. In any case, these tests can be good for showing the extent of viral spread in a community and providing useful public health information and guidance for our overwhelmed healthcare system.
For example (and reminder, I am NOT a healthcare professional but just providing some use cases):
- If you’re symptomatic + IgM positive + IgG negative (or positive): likely active Covid-19 and need treatment plan as determined by a doctor
- If you are asymptomatic + IgM positive + IgG positive: likely a carrier so stay at home for 14 days
- If you are asymptomatic + IgM negative + IgG positive: possibly developed immunity***
*** on immunity, there are interesting implications because there have been proposals on sending these people to the frontline to care for patients.
When will these tests be available at home?
At the time of this post, the FDA has not authorized any test that is available to purchase for testing yourself at home for COVID-19.
Last week, several startups announced that they had cleared regulatory hurdles allowing consumers to collect samples that can be sent in for lab analysis. Unfortunately, this was not the case. In response, earlier this week, the FDA updated their FAQs about COVID-19 testing, stating that at-home sample collecting is not covered under the EUA.
As most of these startups were working on self-collection for RT-PCR tests, they were all affected by this announcement.
Scanwell is not impacted by this ruling as it is bringing a serological test to market. The company is currently working closely with the FDA on guidelines to validate this test in the home. The rapid test has been developed by Innovita, and while there are other manufacturers, Innovita is the only company that detects and displays both IgG and IgM and has NMPA approval (China’s FDA).
You can learn more about how Scanwell will work at-home here.
—
Hopefully, this is a helpful primer from working at the “front of the lines” with one of our portfolio companies. If you have any questions, please feel free to reach out or leave comments.
We realize that the situation is dynamic and many changes happen daily, so we appreciate your understanding as we keep this content up to date. Also, please STAY AT HOME and follow Scanwell on Twitter for updates 🙂
Special thanks to the Scanwell Health team (shout out to Stephen Chen, Dr. Jack Jeng, James hunter and Candace Kim) for teaching me so much, and more importantly, for their incredible leadership at this time. We are all in this together xo

